Is going to be negative one? Well, positive seven. So if you add all that together, you're at negative eight and the whole ion has a negative one. 
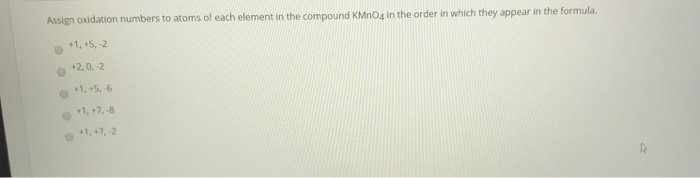
Go with the negative two 'cause it likes to hog two electrons. So I just wanna review that one again because this is a little bit involved. So that means that the manganese has to have a hypothetical charge, an oxidation number of plus seven. And so if each of these four oxygens has a hypothetical charge of negative two, that would be negative eight total and we see that this entire Negative two oxidation number because it likes to hog With oxidation numbers, oxygen is going to have eight It is likely to hog twoĮlectrons and when we think about hypothetical charge But what we generally remember is that oxygen is quite electronegative. Now this one's a little bit more involved to figure out the oxidation numbers. 
And so it's quote hypothetical charge, which isn't so hypothetical in this case, which would be its oxidation Well, each iodine hasĪ negative one charge. Or I suggest you tackle it, is to figure out the oxidation numbers for each of the elementsĪs we go into the reaction, as they are entering the action and as they are exiting the reaction, or I guess you could say onĮither side of the reaction. And the way that I will tackle it, and you might have tackled it If you can figure that out before we work through it together. And what we wanna do in this video is think about which of theĮlements are being oxidized in this reaction and which of the elements are being reduced in this reaction. Have here is a reaction that involves iodine,
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
